| |
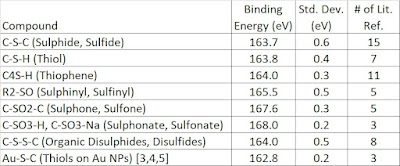
| Table 1. S 2p3/2 binding energies compiled from the NIST database [1] and other sources. |
Notes: 2p3/2 - 2p1/2 doublet separation = 1.18eV, peaks constrained to a 2:1 area ratio (2p3/2 : 2p1/2), generally one sets both peaks to an equal FWHM for ease of use although in pure samples this may not be the case.
Smart et al. [9] and Pratt et al. [10] give an excellent overview of binding energy ranges for the study of mineral surfaces. These ranges can be used with other sulphur containing systems as well. Of particular interest is the assignment for polysulphides eg. (S4)2- = 162.0-163.0 eV, (S5)2- = 161.9 - 163.2 eV, (Sx)2-) = 163.7 eV. Surface species can also play a role in XPS, especially for in-situ fractured sulphide mineral species [11].
[a] Nesbitt et al. [12] give a value of 162.2 eV for the disulphide in arsenopyrite.
[b] A more detailed look at organic sulphur species can be found here.
There is a lot of confusion in the literature when presenting the data for sulphur. Some papers mention S 2p when they really mean S 2p3/2, these are not interchangeable! Please remember to be specific about the exact peak you are referring to.
In a recent paper from Sarah Harmer's group at Flinders University, synchrotron XPS is used to convincingly elucidate surface 3-coordinate, bulk and surface 4-coordinate and bulk 5-coordinate sulfur in the chalcogenide (Fe,Ni)9S8. This work shows that sulfide coordination changes can be seen by XPS [13].
A Na2S2O3.5H2O (sodium thiosulphite cooled to -130C during analysis) reference sample gave S 2p3/2 peak positions of 162.1 eV and 168.1 eV for S*SO3 and SS*O3 moieties, respectively.
A recent paper from Clark et al. [14] highlights how widespread the issue of erroneous peak fitting of S 2p is. Section B within this paper is worth a look as it highlights some of the common errors that should be avoided, these include:
1) Lack of spin–orbit splitting. Doublets (2p3/2 and 2p1/2 peaks) in their appropriate 2:1 ratios, respectively, should be used to represent each chemical state in the material.
2) Inconsistent and widely varying peak widths/full widths at half maximum (FWHMs).
3) Questionable assignments of the peaks to chemical species or oxidation states.
4) Backgrounds that cut through and then extend above the data on the high and low binding energy sides of the peak envelopes.
5) Relatively large range of peak binding energy positions or fit components that are assigned as the same chemical states and should have well defined positions.
6) Noisy spectra, insufficient S/N.
References:
[1] C.D. Wagner, A.V. Naumkin, A. Kraut-Vass, J.W. Allison, C.J. Powell, J.R.Jr. Rumble, NIST Standard Reference Database 20, Version 3.4 (web version) (http:/srdata.nist.gov/xps/) 2003.
[2] Z.E. Pettifer, J.S. Quinton, S.L. Harmer, Minerals Engineering, 184 (2022) 107666.
[2] Z.E. Pettifer, J.S. Quinton, S.L. Harmer, Minerals Engineering, 184 (2022) 107666.
[3] A.N. Buckley, W.M. Skinner, S.L. Harmer, A. Pring, L-J. Fan, Geochimica et Cosmochimica Acta, 73 (2009) 4452-4467.
[4] A.N. Buckley, W.M. Skinner, S.L. Harmer, A. Pring, R.N. Lamb, L.J. Fan, Y. Yang, Canadian Journal of Chemistry, 85 (2007) 767- 781.
[5] S.L. Harmer, A.R. Pratt, H.W. Nesbitt, M.E. Fleet, Canadian Mineralogist, 43 (5) (2005) 1619-1630.
[6] M.E. Fleet, X. Liu, S.L. Harmer, H.W. Nesbitt, Surface Science, 584 (2005) 133-145.
[7] V.P. Zakaznova-Iakovleva, S.L. Harmer, H.W. Nesbitt, G.M. Bancroft, A.R. Pratt, R. Flemming, Surface Science, 600(2) (2006) 348-356.
[8] A.R. Pratt, H.W. Nesbitt, American Mineralogist, 85 (2000) 619-622.
[9] R.St.C. Smart, W.M. Skinner and A.R. Gerson, Surface and Interface Analysis, 28 (1999) 101-105.
[10] A.R. Pratt, I.J. Muir and H.W. Nesbitt, Geochimica et Cosmochimica Acta, 58 (2) (1994) 827-841.
[11] H.W. Nesbitt, M. Scaini, H. Hochst, G.M. Bancroft, A.G. Schaufuss and R. Szargan, American Mineralogist, 85 (2000) 850-857.
[12] H.W. Nesbitt, I.J. Muir, A.R. Pratt, Geochimica et Cosmochimica Acta, 59 (9) (1995) 1773-1786.
[13] Z.E. Pettifer, J.S. Quinton, W.M. Skinner, S.L. Harmer, Applied Surface Science, 504 (2020) 144458.
[14] B.M. Clark, G.H. Major, J.W. Pinder, D.E. Austin, D.R. Baer, M.C. Biesinger, C.D. Easton, S.L. Harmer, A. Herrera-Gomez, A.E. Hughes, W.M. Skinner, M.R. Linford, Journal of Vacuum Science and Technology A, 42 (2024) 063213.