Materials of a graphitic nature (e.g., graphite, graphene, carbon nanotubes etc.) will have a C 1s main peak, attributed to C=C, which can be used as a charge reference set to 284.5 eV. An average of values for graphite from 21 references from the NIST database [1] is 284.46 eV with a standard deviation of 0.14 eV. Note that the well characterized value of 284.5 eV for graphitic carbon is also a strong indicator that this value is not appropriate as a value to use for AdC charge referencing. While these types of samples are generally conductive and if they can be mounted in a manor (in electrical contact with the sample stage) to take advantage of this one should do so. However, many of these types of samples come as a small volume of powders or flakes which are very difficult to mount. Usually, we mount these on a double-sided adhesive which works well but electrically isolates the sample. Oxidation of these types of samples (e.g., graphene oxide) or their functionalization (e.g., functionalized CNTs) can result in them behaving less conductively or as a mixed conductive/insulating material. Samples where these materials are mixed with other conducting or insulating compounds can also result in a mixed conductive/insulating sample. For most of these types of samples we now electrically isolate the sample and charge reference to C 1s at 284.5 eV for the graphitic (C=C) peak.[2]
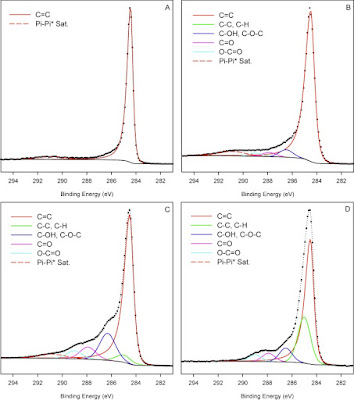
Table 1 from [2] presents general fitting parameters for graphitic, graphene and carbon nanotube type materials. These starting fitting parameters include the main peak asymmetry (defined using an asymmetric Lorentzian (LA) line shape) and π to π* shake-up satellite from a pure graphite standard sample. These fitting parameters are similar to the approach taken by Morgan (Fig. 5, Table 2) [3], Moeini et al. (Table 1) [4], and Gengenbach et al.[5] It is always best to run your own standard (pure graphite, graphene, CNT etc.) to get fitting parameters appropriate for your sample type, instrument and conditions used. Slight differences in the main peak asymmetry and differing shake-up satellite position, shape and intensities are possible for differing classes of graphitic materials. See for example from Morgan[3] where HOPG and nano-onion C 1s spectra show peak-shape differences, likely due to hydrogenation of the sample. However, with this caveat stated, the parameters used based on a graphite standard have worked very well for variety of samples (134) analyzed in the five-year data survey from [2]. Figure 1(A) presents the standard graphite spectrum used to obtain the parameters presented in Table 1. The spectra from Figure 1(B, C and D) show the use of these fitting parameters from Table 1 to effectively model a variety of graphitic component containing materials.
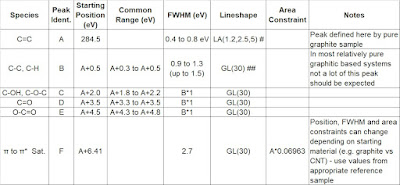
Table 1. General fitting parameters for graphitic/graphene/carbon nanotube type materials. #Line-shape details for CasaXPS. Define asymmetric peak-shape in other software using pure graphite/graphene or CNT sample related your specimens. ##Gaussian/Lorentzian product formula, GL(30) is 30% Lorentzian 70% Gaussian.[2]
Figure 1. Examples of curve-fitting of graphitic type systems using the parameters from Table 1. A) pure graphite, B) carbon nanotube-based material modified in caustic solution, C) oxidized graphene and D) acid modified graphene and organic compound mixture.[2]
References:
[1] C.D. Wagner, A.V. Naumkin, A. Kraut-Vass, J.W. Allison, C.J. Powell, J.R.Jr. Rumble, NIST Standard Reference Database 20, Version 3.4 (web version) (http:/srdata.nist.gov/xps/) 2003.
[2]
M.C. Biesinger, Appl. Surf. Sci. 597 (2022) 153681.[3] D.J. Morgan,
J. Carbon. Res. 7 (2021) 51.
[4] B. Moeini, M.R. Linford, N. Fairley, A. Barlow, P. Cumpson, D. Morgan, V. Fernandez, J. Baltrusaitis.
Surf. Interface Anal. 54 (2022) 67.
[5] T.R. Gengenbach, G.H. Major, M.R. Linford, C.D. Easton,
J. Vac. Sci. Technol. A,
39 (2021) 013204.